
Approximately 17 million dogs suffer from allergic skin disease, including atopic dermatitis, food allergies or flea sensitivity.
Elanco Animal Health Incorporated (NYSE: ELAN) announced the U.S. Food and Drug Administration (FDA) has approved Zenrelia™, a safe, highly effective, and convenient once-daily oral JAK inhibitor for control of pruritus (itching) associated with allergic dermatitis and control of atopic dermatitis in dogs at least 12 months of age.
Itching is one of the top reasons pet owners bring their dog to the veterinarian, and pet owners and veterinarians want more canine dermatology options. Approximately 17 million dogs suffer from allergic skin disease, including atopic dermatitis, food allergies or flea sensitivity. Among pet owners who say their dog’s itch is not under control, 60% say they’ve tried treating the itch, but nothing works.
The approval of Zenrelia represents an important advancement in treating itchy dogs suffering from chronic, acute or seasonal itch and inflammation in a single, once daily tablet from the start. Zenrelia targets itch where it starts by blocking the pathways involved in allergic itch to break the itch-scratch cycle. Zenrelia offers visible improvement from the first dose and minimizes the risk of “rebound itch” which affects many dogs treated with the competitive JAK inhibitor.
“Today is a historic day for Elanco with our first of several expected entries into the fast-growing global canine dermatology market, bringing veterinarians and pet owners a highly effective new solution that got more dogs back to normal levels of itch in a head-to-head study with the current JAK inhibitor on the market1*,” said Jeff Simmons, President and CEO, Elanco Animal Health. “We are excited to offer veterinarians and pet owners a solution that can relieve the burdens of itch, while also becoming just the second animal health company to offer veterinarians a comprehensive portfolio, including parasiticides, vaccines, pain and other therapeutics, and now, dermatology.”
“We’re dedicated to solving these unmet needs in canine dermatology,” said Bobby Modi, Executive Vice President, U.S. Pet Health and Global Digital Transformation. “Zenrelia’s once-daily dosing is more convenient and will be affordable for pet owners with the launch list price about 20% less for nearly all dogs compared to the current JAK inhibitor. Pet owners will appreciate even greater savings in the first 14 days for Zenrelia given its single vs. twice daily dosing. Very importantly, Zenrelia minimizes the opportunity for rebound itch given its consistent once daily dosing compared to the market incumbent.”
The Shelby County Post is a digital newspaper producing news, sports, obituaries and more without a pay wall or subscription needed.


 IDVA offers grant opportunities for non-profit organizations serving the Indiana Veteran Community
IDVA offers grant opportunities for non-profit organizations serving the Indiana Veteran Community
 Gov. Braun activates Indiana National Guard to aid in storm recovery
Gov. Braun activates Indiana National Guard to aid in storm recovery
 Passenger taken to IU-Methodist from two-vehicle Shelbyville crash
Passenger taken to IU-Methodist from two-vehicle Shelbyville crash
 Shelbyville makes major advancement in public safety and emergency response times
Shelbyville makes major advancement in public safety and emergency response times
 Registration open for Tree Trot 5K Run
Registration open for Tree Trot 5K Run
 Flat Rock Christian Church offers shelter Wednesday with potential severe weather
Flat Rock Christian Church offers shelter Wednesday with potential severe weather
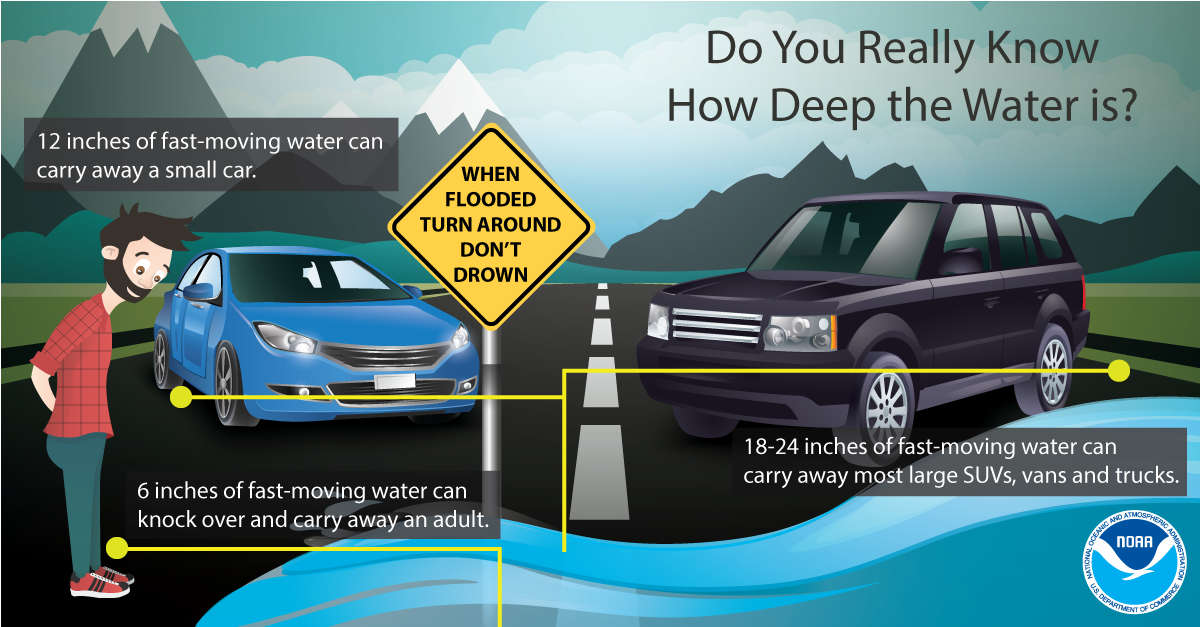 INDOT prepared for severe weather, widespread flooding through weekend
INDOT prepared for severe weather, widespread flooding through weekend




